News
Saving the Hearts of Patients With Chronic Kidney Disease
November 13, 2018
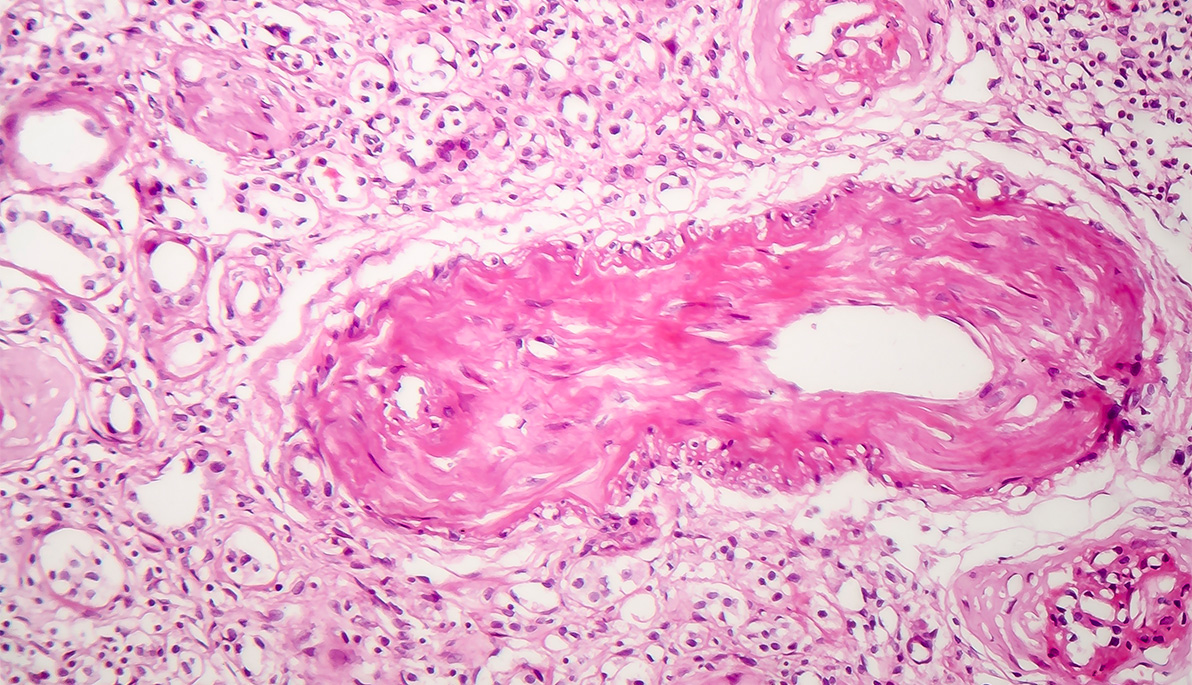
Cardiovascular disease is the most common cause of death for patients with chronic kidney disease. As kidney function decreases, vascular calcification (the accumulation of calcium salts in the tissue of blood vessels) increases and causes arteries to lose their elasticity.
Now, thanks to a $391,041 award from the National Institutes of Health (NIH), Olga V. Savinova, Ph.D., assistant professor of biomedical sciences (NYITCOM), along with an interdisciplinary team of biomedical and biotechnology faculty and students, will examine calcification on the molecular level in order to find a therapy to improve survival rates of patients with chronic kidney disease.
Biomedical scientists believe that the enzyme phosphatase may be the culprit causing increased calcification and hardening of blood vessels. The researchers hypothesize that when the gene for phosphatase, called tissue-nonspecific alkaline phosphatase (TNAP), is overactive, the surplus enzyme produced causes calcification in the arteries. There is currently no standard treatment for vascular calcification, but Savinova expects the study will confirm these theories and lead to a new treatment for heart disease.
“We anticipate our findings will support a new therapy for vascular calcification, a compound known as SBI-425, which robustly inhibits the TNAP enzyme,” said Savinova. “Our long-term goal is to validate the effect of TNAP inhibition and set the stage for testing this compound as a viable pharmacological approach for chronic kidney disease.”
To examine what happens to blood flow in response to increased calcification, Dorinamaria Carka, Ph.D., assistant professor of mechanical engineering in NYIT College of Engineering and Computing Sciences and one of the researchers on the team, will perform computer simulations with the help of an undergraduate engineering student.
“This is a rare opportunity for NYIT engineering students to be involved in an interdisciplinary, computationally intensive research project, involving image processing, multi-physics finite element modeling of biological materials, and processes that aim to resolve the basic science questions as well as provide diagnostic and therapeutic tools for heart disease,” said Carka.
In addition to observing calcification in the coronary arteries of human cadavers, the researchers will also seek to understand TNAP overexpression in mice, specifically whether the surplus phosphatase does indeed contribute to increased calcification. If confirmed, the researchers will then test the potential of a TNAP inhibitor drug in future clinical trials.
The grant’s other investigators include Maria Carrillo Sepulveda, Ph.D., assistant professor of biomedical sciences at NYITCOM, and Maria Plummer, M.D., pathologist and associate professor of clinical specialties at NYITCOM, who will serve as consultants in their areas of expertise.

_Thumb.jpg)


